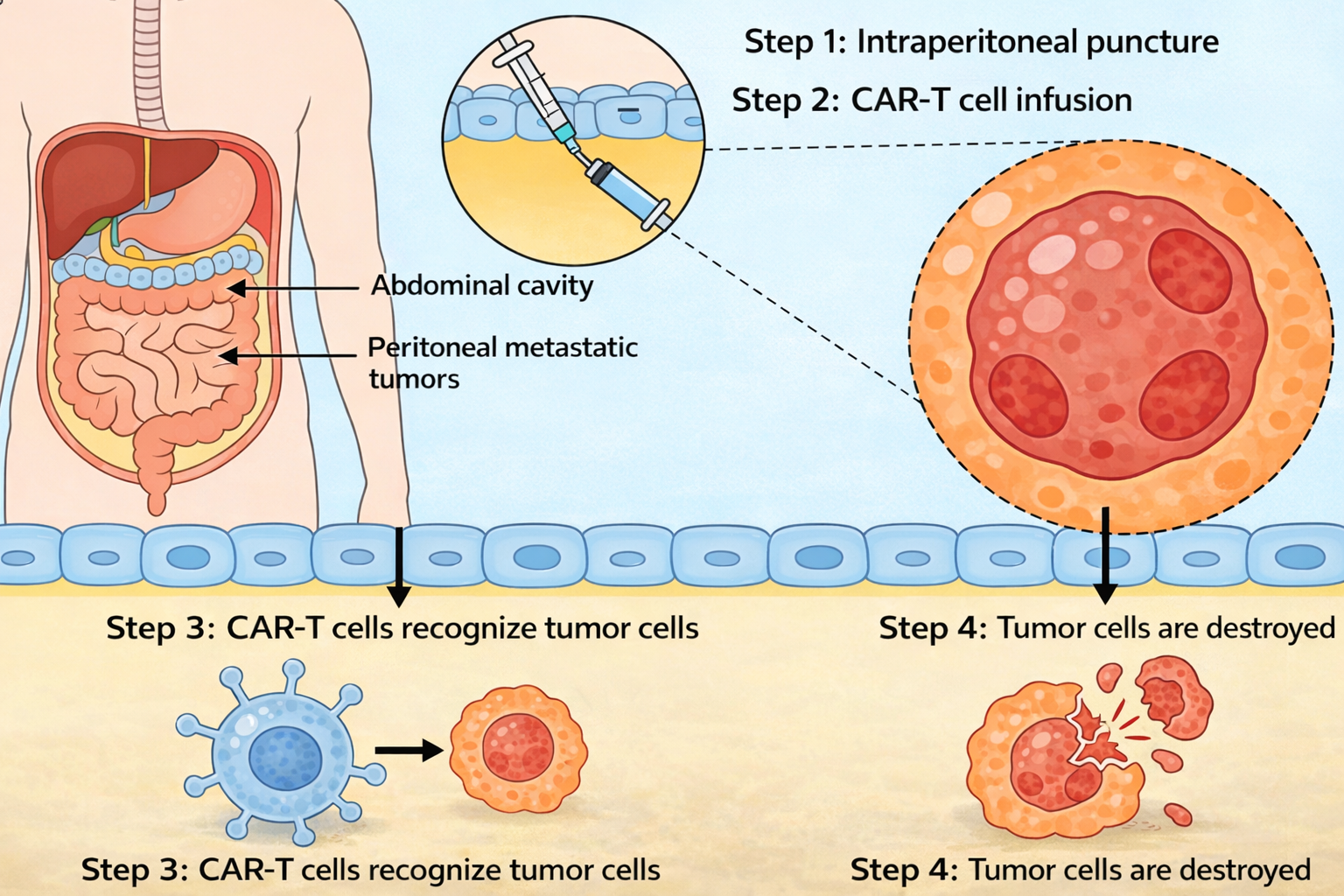
Solid tumors account for approximately 85% of all cancer cases, yet they have long remained a challenging frontier for CAR-T cell therapy. While CAR-T has achieved remarkable success in hematologic malignancies, its performance in solid tumors has been limited. Tumor heterogeneity, an immunosuppressive microenvironment, and off-target toxicity collectively represent major barriers to its clinical application.
In February 2026, a Chinese research team made a significant breakthrough in this field. The hypoxia-responsive CEA-targeted CAR-T cell therapy (PC13), jointly developed by the First Affiliated Hospital of Zhejiang University School of Medicine and Chongqing Precision Biotech Co., Ltd., was published in Nature Cancer. This study demonstrated, for the first time in humans, the feasibility of a “hypoxia-gated” CAR-T strategy, offering a new therapeutic approach for patients with advanced CEA-positive solid tumors.
Challenges of CAR-T Therapy in Solid Tumors
Compared with hematologic cancers, solid tumors exhibit far greater biological complexity, which directly impacts the effectiveness of CAR-T therapy.
Tumor heterogeneity leads to uneven antigen expression across different cell populations, making it difficult for CAR-T cells to maintain consistent cytotoxic activity. At the same time, the tumor microenvironment—characterized by hypoxia, immunosuppressive cells, and inhibitory factors—can significantly impair T cell function and persistence.
From a safety perspective, low-level expression of target antigens in normal tissues may result in off-target effects, posing additional clinical risks.
📌 Key challenges of CAR-T therapy in solid tumors include:
1️⃣ Heterogeneous antigen expression leading to inconsistent efficacy
2️⃣ Immunosuppressive microenvironment limiting T cell activity
3️⃣ Increased risk of off-target toxicity
4️⃣ Insufficient T cell infiltration into tumor tissuesThese factors together form the core bottleneck in advancing CAR-T therapy for solid tumors.
PC13 Design: Environment-Responsive Precision Activation
The design of PC13 centers on “tumor-specific activation,” integrating CAR expression with the hypoxic tumor microenvironment.
Under normoxic conditions, CAR expression remains low. Once CAR-T cells enter tumor tissues, hypoxia induces upregulation of CAR expression, enabling selective activation within the tumor site. This “hypoxia-gated” mechanism enhances spatial specificity.
CEA (carcinoembryonic antigen) serves as the target antigen. It is highly expressed in multiple solid tumors, including colorectal cancer, gastric cancer, and certain lung cancers, providing a clear targeting basis.
To address different clinical scenarios, two administration routes were designed:
● Intraperitoneal infusion: suitable for peritoneal metastases or localized tumors
● Intravenous infusion: suitable for systemic tumor distribution
This differentiated approach helps improve CAR-T cell accumulation at tumor sites.
Clinical Results: Efficacy Signals and Safety Profile
In a Phase I clinical trial, 43 patients with advanced CEA-positive solid tumors who had failed multiple prior therapies were enrolled.
Patients were divided into:
● Intraperitoneal group (17 patients)
● Intravenous group (26 patients)
Some patients showed significant tumor shrinkage. In one case, the sum of target lesion diameters decreased by 75.98%, with complete disappearance of one lesion at 6-month follow-up. Among patients achieving partial response, the median progression-free survival (mPFS) reached 6.3 months, showing an advantage over traditional later-line therapies.
📊 Key clinical findings:
● Significant tumor shrinkage (>70%) in some patients
● Complete radiological disappearance of certain lesions
● mPFS of 6.3 months, exceeding conventional expectations
● Variations in efficacy between administration routes
In terms of safety, most adverse events were mild to moderate. Cytokine release syndrome (CRS) and neurotoxicity were generally manageable, with no severe uncontrolled events observed.
Technological Significance: From Passive Targeting to Active Regulation
The value of PC13 lies not only in its clinical efficacy but also in its innovative technological approach.
Traditional CAR-T therapies rely on constant CAR expression. In contrast, PC13 introduces an environment-responsive mechanism, allowing CAR activity to dynamically adapt to the tumor microenvironment. This design helps balance efficacy and safety.
Key innovations include:
● Shifting CAR expression from constant to conditionally triggered
● Leveraging tumor hypoxia for spatial selectivity
● Enhancing local anti-tumor effects while reducing systemic toxicity
This strategy opens new directions for optimizing CAR-T therapy in solid tumors and lays the groundwork for next-generation “smart” cell therapies.
Challenges and Future Optimization
Despite encouraging results, further development of PC13 requires continued optimization. Enhancing T cell infiltration into solid tumors remains critical for improving efficacy consistency. The hypoxia-responsive system also needs refinement to ensure reproducibility across patients.
Industrial challenges must also be addressed, including manufacturing timelines, quality control, and cost management, all of which impact clinical scalability.
Future research directions may include:
● Improving T cell infiltration and persistence
● Refining hypoxia-responsive control mechanisms
● Exploring combination therapies (e.g., checkpoint inhibitors)
● Advancing standardized production and cost reduction
Conclusion
Hypoxia-responsive CEA-targeted CAR-T therapy (PC13) represents a promising and innovative approach in solid tumor immunotherapy. By integrating cellular function with tumor microenvironment characteristics, this strategy establishes a new balance between safety and efficacy.
Current clinical data suggest that PC13 demonstrates meaningful anti-tumor activity with a manageable safety profile. As research progresses and technology continues to evolve, this therapy may play an increasingly important role in solid tumor treatment and serve as a key reference for CAR-T applications in complex cancers.
With the support of global platforms such as Dengyue Pharma, accessibility to innovative therapies is steadily improving. As more advanced technologies mature, immunotherapy for solid tumors may finally enter a true breakthrough era.